New Medical Ideas
- Home
- Introduction
- Examples of New Medical Innovations
- Smartphone Medical Applications
- Recent innovative medical ideas
- New Potential Drugs
- Links
- Share Your Ideas
- Intellectual Property Protection
- Approval Process
- Careers in Medical Research
- Mysterious diseases
- Disclaimer
- Healthcare Costs by Age and Disease
- Medical Devices from Israel
- Contact Form
Marketing and Approval of New Ideas
How do you bring your idea to the market?
What happens between the time when you got the idea, and when you get the Nobel Prize in Medicine?
Bringing your idea to the market includes finding resources to fund your research and development, file patents, establish relations with medical and/or pharmaceutical companies, and determining the value and marketability of your innovative idea.
In addition, you must of course show that your idea actually does what it is assumed to do; heal someone. This has to be proven with meticulous medical research. But such research is no simple matter. It doesn’t start and end with giving two patients a drug or a placebo. After you have seen that it worked once, you have to replicate it many times under different conditions, and then publish the results in some respectable medical journal. Only thereafter, might it be possible to ask for approval from the FDA if it proves to have any worth, before it is possible to actually launch it.
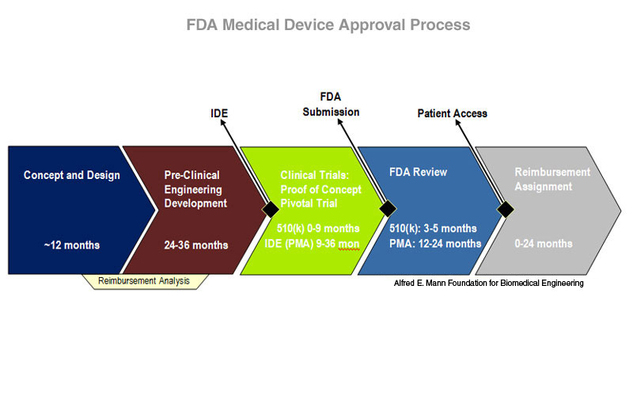
The Process of Getting New Medical Ideas Approved
Understanding Clinical Trials
Choosing to participate in a clinical trial is an important personal decision. The following frequently asked questions provide detailed information about clinical trials. In addition, it is often helpful to talk to a physician, family members, or friends about deciding to join a trial. After identifying some trial options, the next step is to contact the study research staff and ask questions about specific trials. Read more (from NIH).
Human Clinical Trial Process
Phase I studies assess the safety of a drug or device. This initial phase of testing, which can take several months to complete, usually includes a small number of healthy volunteers (20 to 100), who are generally paid for participating in the study. The study is designed to determine the effects of the drug or device on humans including how it is absorbed, metabolized, and excreted. This phase also investigates the side effects that occur as dosage levels are increased. About 70% of experimental drugs pass this phase of testing.
Phase II studies test the efficacy of a drug or device. This second phase of testing can last from several months to two years, and involves up to several hundred patients. Most phase II studies are randomized trials where one group of patients receives the experimental drug, while a second "control" group receives a standard treatment or placebo. Often these studies are "blinded" which means that neither the patients nor the researchers know who has received the experimental drug. This allows investigators to provide the pharmaceutical company and the FDA with comparative information about the relative safety and effectiveness of the new drug. About one-third of experimental drugs successfully complete both Phase I and Phase II studies.
Phase III studies involve randomized and blind testing in several hundred to several thousand patients. This large-scale testing, which can last several years, provides the pharmaceutical company and the FDA with a more thorough understanding of the effectiveness of the drug or device, the benefits and the range of possible adverse reactions. 70% to 90% of drugs that enter Phase III studies successfully complete this phase of testing. Once Phase III is complete, a pharmaceutical company can request FDA approval for marketing the drug.
Phase IV studies, often called Post Marketing Surveillance Trials, are conducted after a drug or device has been approved for consumer sale. Pharmaceutical companies have several objectives at this stage: (1) to compare a drug with other drugs already in the market; (2) to monitor a drug's long-term effectiveness and impact on a patient's quality of life; and (3) to determine the cost-effectiveness of a drug therapy relative to other traditional and new therapies. Phase IV studies can result in a drug or device being taken off the market or restrictions of use could be placed on the product depending on the findings in the study.
Phase II studies test the efficacy of a drug or device. This second phase of testing can last from several months to two years, and involves up to several hundred patients. Most phase II studies are randomized trials where one group of patients receives the experimental drug, while a second "control" group receives a standard treatment or placebo. Often these studies are "blinded" which means that neither the patients nor the researchers know who has received the experimental drug. This allows investigators to provide the pharmaceutical company and the FDA with comparative information about the relative safety and effectiveness of the new drug. About one-third of experimental drugs successfully complete both Phase I and Phase II studies.
Phase III studies involve randomized and blind testing in several hundred to several thousand patients. This large-scale testing, which can last several years, provides the pharmaceutical company and the FDA with a more thorough understanding of the effectiveness of the drug or device, the benefits and the range of possible adverse reactions. 70% to 90% of drugs that enter Phase III studies successfully complete this phase of testing. Once Phase III is complete, a pharmaceutical company can request FDA approval for marketing the drug.
Phase IV studies, often called Post Marketing Surveillance Trials, are conducted after a drug or device has been approved for consumer sale. Pharmaceutical companies have several objectives at this stage: (1) to compare a drug with other drugs already in the market; (2) to monitor a drug's long-term effectiveness and impact on a patient's quality of life; and (3) to determine the cost-effectiveness of a drug therapy relative to other traditional and new therapies. Phase IV studies can result in a drug or device being taken off the market or restrictions of use could be placed on the product depending on the findings in the study.
Financing
Obtaining financing for new medical ideas is always a challenge for innovators. Help is often needed to advance device and technology innovations to interest investors. With the difficult economy and increasing pressure to minimize risk, few investors or potential partners are willing to consider new medical device concepts without working prototypes that have undergone rigorous development and pre-clinical testing.